 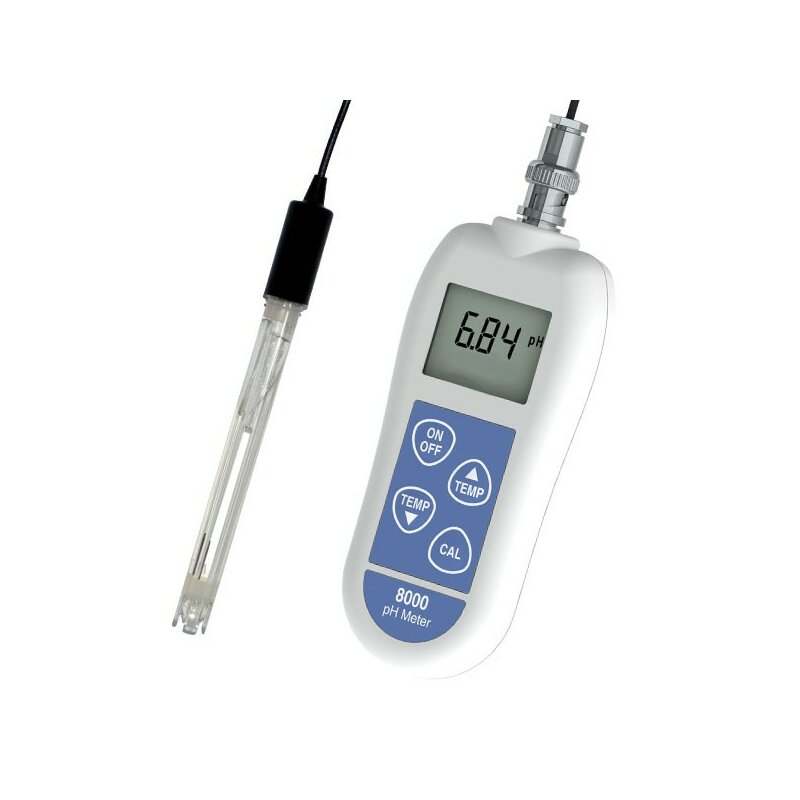
The pH-sensitive glass membrane is generally spherical to simplify the manufacture of a uniform membrane. Glass is a solid electrolyte, for which alkali-metal ions can carry current. : 125 However, certain aspects of design are published. pH electrode and reference electrode design ĭetails of the fabrication and resulting microstructure of the glass membrane of the pH electrode are maintained as trade secrets by the manufacturers. This equilibration provides a stable pH measurement. To measure the pH of a solution, the electrodes are used as probes, which are dipped into the test solutions and held there sufficiently long for the hydrogen ions in the test solution to equilibrate with the ions on the surface of the bulb on the glass electrode. The pH meter is calibrated with solutions of known pH, typically before each use, to ensure accuracy of measurement. A detailed description of combination electrodes is given in the article on glass electrodes. įor simplicity, many pH meters use a combination probe, constructed with the glass electrode and the reference electrode contained within a single probe. All other potential differences in the circuit do not vary with pH and are corrected for by means of the calibration. The voltage varies from test solution to test solution depending on the potential difference created by the difference in hydrogen-ion concentrations on each side of the glass membrane between the test solution and the solution inside the glass electrode. The circuit can be thought of as going from the conductive element of the reference electrode to the surrounding potassium-chloride solution, through the ceramic membrane to the test solution, the hydrogen-ion-selective glass of the glass electrode, to the solution inside the glass electrode, to the silver of the glass electrode, and finally the voltmeter of the display device. On immersion of the glass electrode and the reference electrode in the test solution, an electrical circuit is completed, in which there is a potential difference created and detected by the voltmeter. The display consists of a voltmeter, which displays voltage in units of pH. This conductor is immersed in an electrolyte solution, typically potassium chloride, which comes into contact with the test solution through a porous ceramic membrane. The reference electrode is insensitive to the pH of the solution, being composed of a metallic conductor, which connects to the display. The magnitude of the electrochemical potential across the glass bulb is linearly related to the pH according to the Nernst equation. The electronic amplifier detects the difference in electrical potential between the two electrodes generated in the measurement and converts the potential difference to pH units. On immersion in the solution to be tested, hydrogen ions in the test solution exchange for other positively charged ions on the glass bulb, creating an electrochemical potential across the bulb. The glass electrode for measuring the pH has a glass bulb specifically designed to be selective to hydrogen-ion concentration. The design of the electrodes is the key part: These are rod-like structures usually made of glass, with a bulb containing the sensor at the bottom. The electrodes, or probes, are inserted into the solution to be tested. It usually has a glass electrode and a reference electrode, or a combination electrode. They comprise a simple electronic amplifier and a pair of electrodes, or alternatively a combination electrode, and some form of display calibrated in pH units. Potentiometric pH meters measure the voltage between two electrodes and display the result converted into the corresponding pH value. Using an early Beckman pH meter in a lab Principle of operation These have tips suitable for piercing semi-solids, have electrode materials compatible with ingredients in food, and are resistant to clogging. In addition to measuring the pH of liquids, specially designed electrodes are available to measure the pH of semi-solid substances, such as foods. The devices have been miniaturized, enabling direct measurement of pH inside of living cells. Īdvances in the instrumentation and in detection have expanded the number of applications in which pH measurements can be conducted. 
pH meters are used for soil measurements in agriculture, water quality for municipal water supplies, swimming pools, environmental remediation brewing of wine or beer manufacturing, healthcare and clinical applications such as blood chemistry and many other applications. Knowledge of pH is useful or critical in many situations, including chemical laboratory analyses. The rate and outcome of chemical reactions taking place in water often depends on the acidity of the water, and it is therefore useful to know the acidity of the water, typically measured by means of a pH meter. 2.2 pH electrode and reference electrode design. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
